|
https://ift.tt/2To0Got
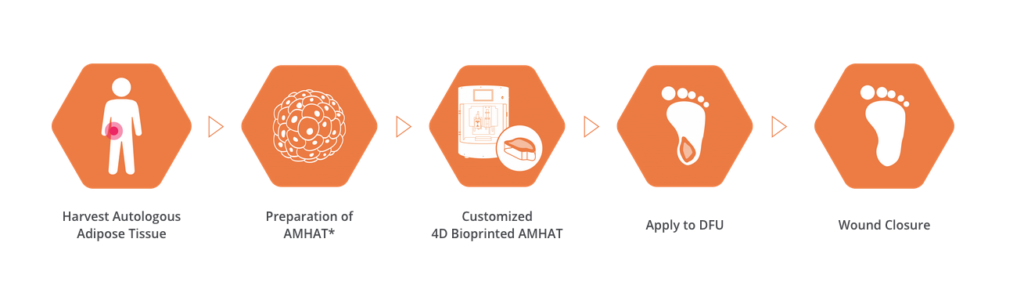
ROKIT Healthcare’s bioprinting-based diabetic foot treatment kit registered as a U.S. FDA medical device https://ift.tt/3uhvvYw ROKIT Healthcare has completed registration of its Dr. INVIVO-based diabetic foot treatment with the U.S. FDA and has successfully completed clinical studies in the U.S. Since 2019, the company has conducted clinical studies in India, Korea and Turkey, and based on the most recent study results in the U.S., the treatment has shown promising results in all of the study patients with no side effects. The company reported that the study subjects were patients of chronic diabetic foot ulcer with an average of 64.4 years of age, 15 years of diabetes duration, and obesity in 90% of the patients. The principal investigator behind the study is Dr. David G. Armstrong, PhD, DPM, who is Professor of Clinical Surgery at University of Southern California and has produced more than 510 peer-reviewed research papers in scholarly medical journals and 90 book chapters with expertise in the treatment and clinical care of the diabetic foot. Dr. Armstrong is past Chair of Scientific Sessions for the American Diabetes Association’s Foot Care Council and the founder and co-chair of the International Diabetic Foot Conference, the largest annual international symposium on the diabetic foot in the world.
ROKIT Healthcare’s diabetic foot ulcer treatment, which consists of the printer Dr. INVIVO DFU and patient-specific autologous solution extraction kits, uses 3D scanning and AI-based interpretation of patient wounds to create customized skin regeneration patches.
Printing via 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing https://3dprint.com May 26, 2021 at 06:36AM
0 Comments
Leave a Reply. |
Categories
All
Archives
April 2023
|


 RSS Feed
RSS Feed
