|
OPM Licensed for Marketing & Manufacturing 3D Printed Medical Devices in Asia https://ift.tt/2AipFiI
OPM was just recently accredited as a foreign medical device manufacturer by the Japanese Ministry of Health, Labour, and Welfare. With this international development, they will also be creating OPM Asia in partnership with JSR Corp. of Tokyo. JSR is known for their strength in the life sciences industry in Asia, including a personalized medicine ecosystem, 3D software, drug discovery, and more. The license will allow the new partners to market and manufacture medical devices in the following countries, with products expected to be on the Asian market by 2019 in:
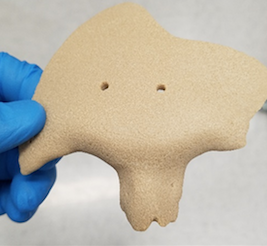
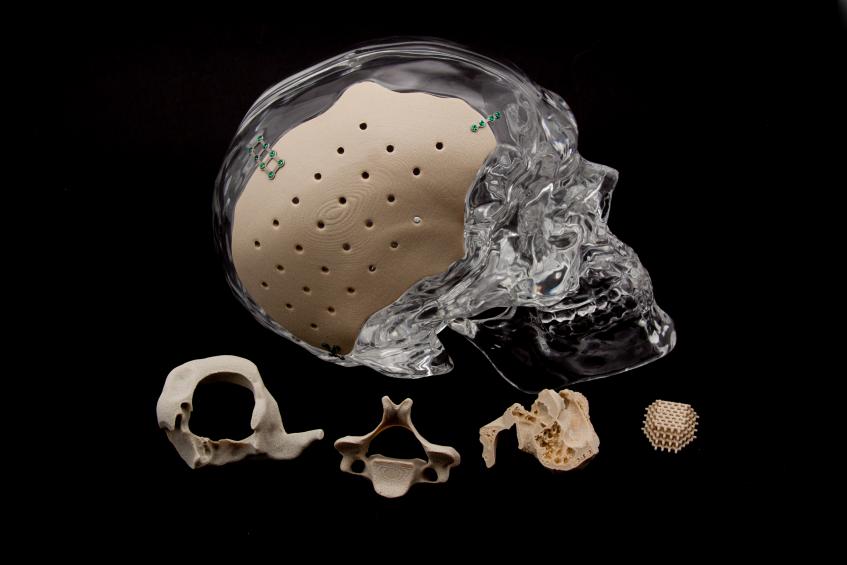
With their ongoing use of PEKK, the OPM team produces implants very similar to bone that are osteoconductive and encourage the growth of natural bone once implanted. It is also radiolucent—meaning that it works well for patients having MRIs as well.
OPM offers comprehensive services around the world, also serving as an OEM and regulatory filer. When PEKK was originally created by DuPont, OPM was approved to make their own OXPEKK material, and since then they have patented OXPEKK LTS for 3D printing medical implants.
With the OPM and JSR partnership in place, they also intend to expand into other areas:
What do you think of this news? Let us know your thoughts! Join the discussion of this and other 3D printing topics at 3DPrintBoard.com. [Source: MDDI; Images: OPM] Printing via 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing https://3dprint.com July 30, 2018 at 03:09AM
0 Comments
Leave a Reply. |
Categories
All
Archives
April 2023
|


 RSS Feed
RSS Feed
