|
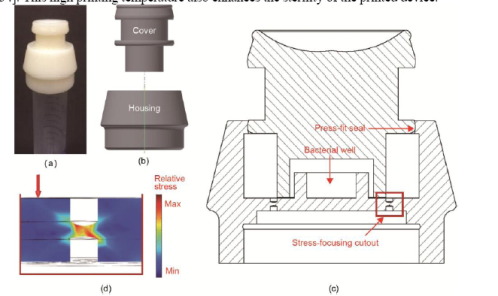
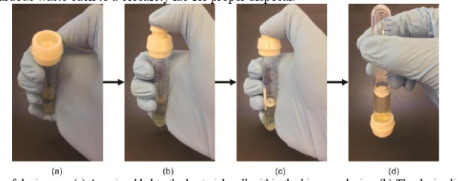
3D Printed Device Allows Living Biosensor to be Deployed in the Field http://bit.ly/2ERvVPt Genetically modified organisms, or GMOs, are subject to strict regulation. In a paper entitled “An Additive Manufacturing Approach that Enables the Field Deployment of Synthetic Biosensors,” a group of researchers explains that living biosensors, which can be deployed for disease diagnosis in the field, are considered GMOs and are therefore subject to that same regulation. Using 3D printing is one way in which these biosensors can be made secure and acceptable for deployment in the field. In rural and less-developed areas, the researchers explain, the pathogen P. aeruginosa can be acquired in local clinics. The organism is found in lung sputum samples upon infection, and the samples can be collected and placed in commercially available sample tubes. The researchers’ study focuses on creating a biosensor that could be used to immediately diagnose the disease in the field. They used E. coli as a biosensor, but needed a way to safely transport the dangerous bacteria from the laboratory. So they 3D printed a unique cap that completely encases the bacteria and can be securely sealed onto a sample tube containing lung sputum samples.
P. aeruginosa, they continue, functions as an opportunistic pathogen in the lungs of patients with cystic fibrosis. Currently, it is diagnosed either with enzyme-linked immunosorbent assays (ELISAs) and real-time polymerase chain reaction (PCR) assays, which are either expensive or require extensive training to administer. The researchers’ hope is that their biosensor, with its secure 3D printed containment device, can be used as an inexpensive, easy-to-use alternative. The 3D printed device was designed in Autodesk Inventor and 3D printed in ABS ULTRAT using a Zortrax M200 3D printer.
The biosensor quickly senses the presence of the P. aeruginosa pathogen, and because of its enclosure within the 3D printed device, it should face far fewer regulatory hurdles, allowing it to be deployed freely outside the lab.
Authors of the paper include Daniel Wolozny, John R. Lake, Paul G. Movizzo, Zhicheng Long and Warren C. Ruder. Discuss this and other 3D printing topics at 3DPrintBoard.com or share your thoughts below.
Printing via 3DPrint.com | The Voice of 3D Printing / Additive Manufacturing https://3dprint.com December 26, 2018 at 11:49AM
0 Comments
Leave a Reply. |
Categories
All
Archives
April 2023
|


 RSS Feed
RSS Feed
